Tag: side-effects
USA: Senator Ron Johnson holds round table discussion with those suffering from coronavirus vaccine side-effects
“What’s been happening over the last year and a half is, people that are trying to tell the truth, people that are trying to get answers, are having a hard time getting their truths communicated, without being vilified, without being ridiculed, without being censored.” More...
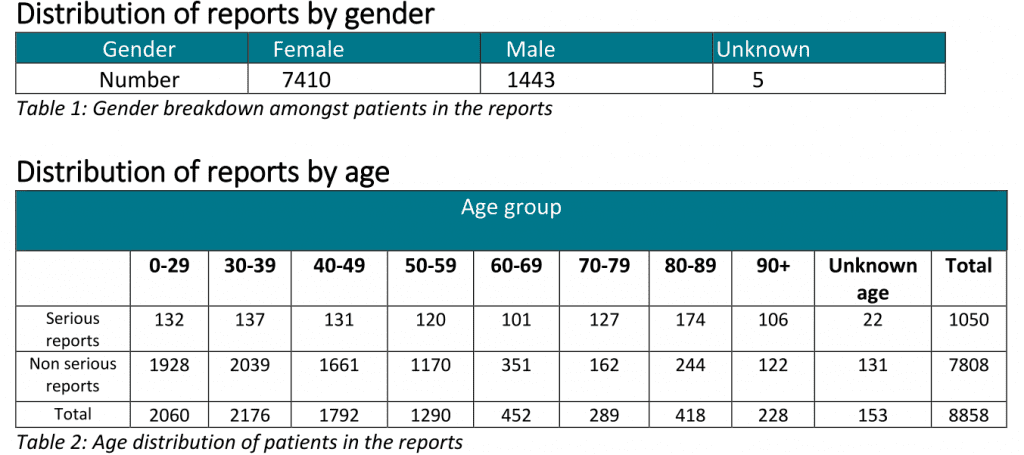
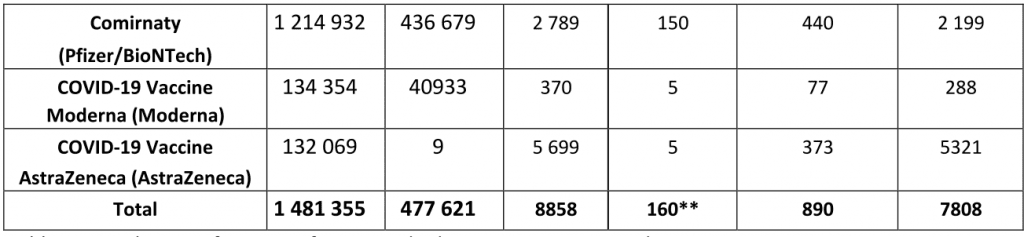
Norway: 1.2 million coronavirus vaccinations, 160 deaths, 890 serious side-effects, 7,800 other side effects
Norway vaccine side-effects to 11th May 2021 (Note: The Astrazeneca Vaxevria vaccine is no longer used in Norway)
Norwegian Medicines Agency report
UK: Eric Clapton had a ‘disastrous reaction’ to AstraZeneca coronavirus vaccination
Photo by Mufid Majnun on Unsplash More...
UK: Princess Michael of Kent is being treated for blood clots after receiving two doses of the AstraZeneca vaccine
Princess Michael of Kent is being treated for blood clots after receiving two doses of the Oxford/AstraZeneca vaccine – although no link has been established. More...
UK: AstraZeneca coronavirus vaccine blood clot cases double to 168 in two weeks, 32 deaths reported
The risk of a serious blood clot from AstraZeneca jab has doubled in a fortnight, new data show. More...
Thailand: seven medical staff suffered partial paralysis after receiving Sinovac coronavirus vaccine
Seven people suffered from partial paralysis after receiving the Sinovac vaccine, a doctor from Chulalongkorn University said. More...
USA: CDC is recommending that people who experience certain new symptoms after receiving Johnson & Johnson’s coronavirus vaccine seek immediate medical treatment
The Centers for Disease Control and Prevention is recommending that people who experience certain new symptoms after receiving Johnson & Johnson’s Covid-19 vaccine seek immediate medical treatment. More...
Denmark: 29 claims for compensation made for side-effects following AstraZeneca coronavirus vaccination
Denmark has received the first claim for compensation for a death caused by a special type of blood clot, which the authorities link with the Astrazeneca vaccine.
CDC: Moderna’s COVID-19 coronavirus vaccine could lead to more side effects than Pfizer/BioNTech’s
New research from the U.S. Centers for Disease Control and Prevention suggests that people who receive Moderna’s COVID-19 vaccine might experience more side effects than those who get Pfizer/BioNTech’s vaccine. More...
Sweden: Almost six times as many suspected side effects reported for AstraZeneca’s vaccine compared to Pfizer and Moderna
The Swedish Medical Products Agency has received almost six times as many reports of suspected side effects for AstraZeneca’s vaccine, compared with the other two EU-approved vaccines Pfizer/Biontech and Moderna, in relation to the number of people vaccinated. More...
Virginia man suffers ‘rare’ severe reaction to coronavirus vaccine
A Virginia man suffered a severe reaction after getting his COVID-19 vaccine and doctors believe it was a direct result of the shot. More...