Category: Denmark
Denmark: Over 71,000 Covid-19 vaccine adverse events reported
Hundreds of Danes have received compensation for Covid-19 vaccine injuries. More...
Study: Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine
An investigation by five German scientists of the variability of batches of the Pfizer/BionTech BNT162b2 mRNA COVID-19 vaccine, using a recent Danish study as the basis for their study. More...
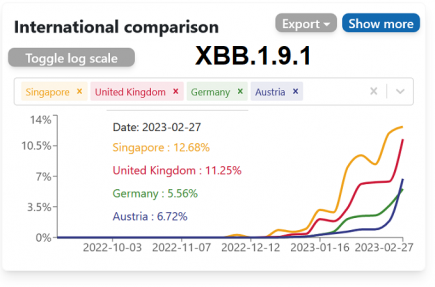
XBB.1.9.1 – a new variant of concern? – 4 updates
XBB.1.9.1, a new recombinant subvariant that is suddenly cropping up across the globe, is causing some concern amongst epidemiologists. More...
A huge wave of Covid-19 deaths is rolling across the globe *1 Update*
A huge winter wave of Covid deaths is spreading across the world, almost entirely unreported by the mainstream media. More...
Record Covid deaths in Japan, pneumonia increases in Italy
Just two days ago, the WHO warned of an increase in Covid deaths across the globe. More...
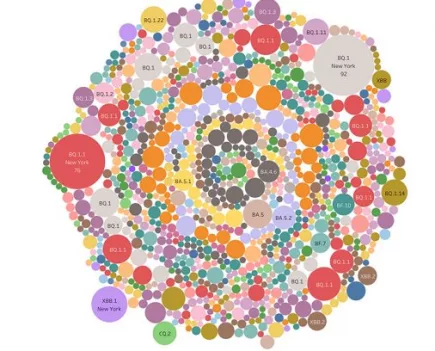
Denmark’s Covid variant soup is turning into a Covid variant smoothie
When we last checked in October 2022, Denmark had sequenced 120 different Covid variants – this month, they have sequenced 249 Covid variants. More...
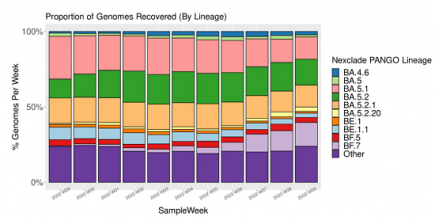
BF.7: Another contender for global domination
The XBB and BQ.1 variants have recently been making headlines, but another SARS-CoV-2 variant, BF.7 has been closely tracking their success across the globe. More...
Denmark: A soup of 120 Covid variants sequenced in one month
You may have heard the expression “a soup of Covid variants” recently.. More...
The Queen of Denmark tests positive for Covid-19 after THAT funeral
Queen Margrethe of Denmark has tested positive for Covid-19 for the second time in one year after attending the biggest Covid superspreader event in pandemic history – the funeral of Queen Elizabeth II in Great Britain. More...
Denmark abandons Covid-19 vaccinations for under-18s
“For children and young people under the age of 18, from 1 July 2022 it will no longer be possible to get the 1st injection, and from 1 September 2022 it will no longer be possible to get the 2nd injection.” More...