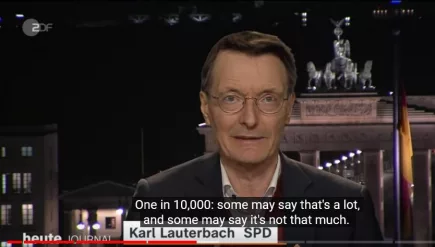Category: Germany
Germany: One man, 217 Covid vaccinations
Lancet study: ‘Here, we report on a 62-year-old male hypervaccinated individual from Magdeburg, Germany, who deliberately and for private reasons received 217 vaccinations against SARS-CoV-2 within a period of 29 months.’ More...
Study: Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine
An investigation by five German scientists of the variability of batches of the Pfizer/BionTech BNT162b2 mRNA COVID-19 vaccine, using a recent Danish study as the basis for their study. More...
Prof Arne Burkhardt: 2nd Conference on Vaccine Adverse Events
Another heart-stopping conference with Professor Arne Burkhardt, looking at bizarre new clotting pathologies found in post-mortem examinations of patients carried out in Germany. More...
Germany: One in 10,000 vaccinated for Covid are severely injured by the jab
Germany’s Health Minister, Karl Lauterbach, has stated in an interview that around one person in ten thousand is severely injured as a result of a Covid-19 vaccination. More...
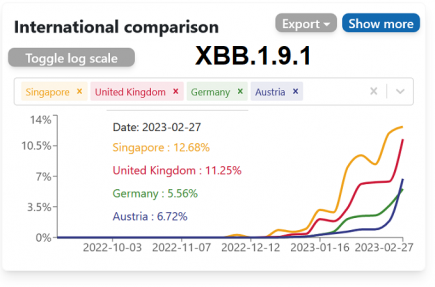
XBB.1.9.1 – a new variant of concern? – 4 updates
XBB.1.9.1, a new recombinant subvariant that is suddenly cropping up across the globe, is causing some concern amongst epidemiologists. More...
Covid reinfection causes ‘an immune deficiency that can no longer be cured’
The German Health Minister seems to have broken the global omertà about the dangers of Covid reinfections yesterday by saying “those affected are often dealing with an immune deficiency that can no longer be cured”. More...
Record Covid deaths in Japan, pneumonia increases in Italy
Just two days ago, the WHO warned of an increase in Covid deaths across the globe. More...
Europe: Another record-breaking Covid winter wave predicted
These European Covid-19 infection charts do NOT look good – almost every country in Europe has a higher Covid case level now than before the huge Omicron winter wave of 2021. More...
Germany: Chancellor Olaf Scholz and Interior Minister Nancy Faeser test positive for Covid
German Chancellor Olaf Scholz has tested positive for COVID-19. More...
Long Covid: Petition calls on Germany to expedite BC007 clinical trials
A new petition on Change.org calls on the German government to expedite clinical trials for BC007, a promising potential treatment for Long COVID. More...