Category: Reinfection
UK NHS healthcare workers launch group litigation against government for Covid-19 injuries received at work
UK NHS workers are preparing to take legal action against the British government for failing to protect them during the Covid-19 pandemic. More...
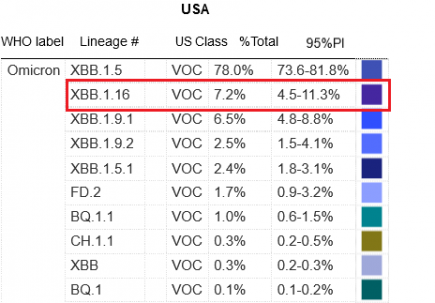
XBB.1.16 is the second most common variant in the US
XBB.1.16 has debuted on the CDC Covid variant list today in SECOND PLACE. More...
Covid reinfection causes ‘an immune deficiency that can no longer be cured’
The German Health Minister seems to have broken the global omertà about the dangers of Covid reinfections yesterday by saying “those affected are often dealing with an immune deficiency that can no longer be cured”. More...
A huge wave of Covid-19 deaths is rolling across the globe *1 Update*
A huge winter wave of Covid deaths is spreading across the world, almost entirely unreported by the mainstream media. More...
UK: Keep kids at home if they are sick, adults should wear a mask
UKHSA: Keep kids at home if they are sick, unwell adults should wear a facemask.
More...XBB.1.5 at a staggering FORTY PERCENT of sequences in the US
These numbers were so shocking that, when they were leaked online yesterday, we didn’t believe them. More...
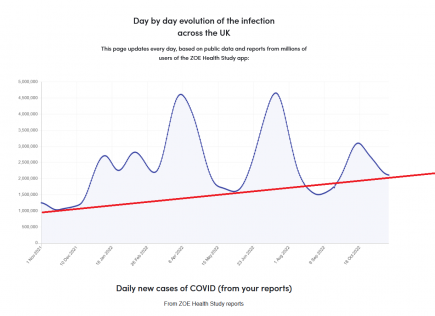
UK: Covid on the rise in FIFTH wave of the year
The UK ZOE app is showing Covid cases are once again on then rise in what would be the fifth wave of 2022 in the UK. More...
COVID-19 reinfections increase risk of organ failure, death
New research published in Nature Medicine magazine shows the devastating damage that can be caused to the human body by Covid-19 reinfections, with each reinfection increasing the risk, regardless of vaccination status. More...
Japan: new Covid wave begins with BF.5 as leading variant again
Japan is seeing a new wave of coronavirus rolling in, just weeks after the last devastating wave tailed off. More...
XBB set to become the dominant variant in Singapore
XBB is set to become the dominant variant in Singapore, and is already dominant in Bangladesh. More...