Category: Norway
A huge wave of Covid-19 deaths is rolling across the globe *1 Update*
A huge winter wave of Covid deaths is spreading across the world, almost entirely unreported by the mainstream media. More...
Norway: 46% of Covid infected report long-term effects
“As many as 46 per cent of those who had been infected with Covid in our study report long-term effects.” More...
ECDC & WHO: Joint statement on hepatitis in kids
The World Health Organisation have issued a joint statement with the ECDC on the recent cases of hepatitis in children. More...
West European summer Covid wave brewing *6 updates, 1 prediction*
A new western European summer Covid wave appears to be developing, with increases in cases in countries that also saw large waves in the summer of 2021. More...
Norway: Covid infections by vaccination status in 2022 *1 UPDATE*
Norway has released its latest report on Covid infections by vaccination status, so we will be keeping track of those figures in this thread. More...
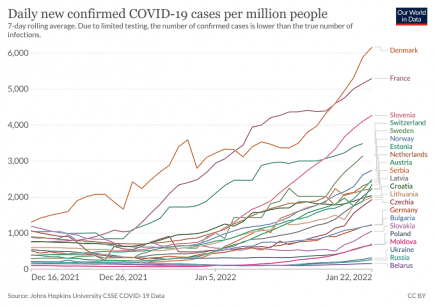
Europe: Another huge Covid wave is building right across the continent
Almost every country in Europe is now seeing a big increase in Covid cases. More...
Norway: 248 deaths and nearly 52,000 reports of other Covid vaccine side-effects
From a population of just 5.3 million people, Norway has reported a total of 248 deaths as suspected side effects after a Covid vaccine, according to the latest Adverse Reaction Report from the Norwegian Medicines Agency, published on Thursday, 20th January. More...
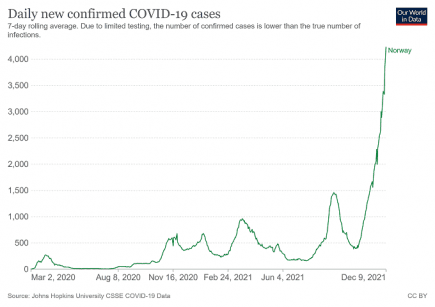
Norway: new Covid restrictions introduced as highest ever hospitalizations recorded
UPDATE: Norway has the highest infection rate by in Europe. More...
Omicron in Norway: a warning to the world
Norway’s latest Covid figures show soaring numbers of infections and hospital inpatients, with big increases in both ICU and ventilated patients. More...
Omicron: 73% attack rate at Oslo restaurant superspreader event
As of 8th December, around 70 % of just over 100 participants at a Christmas party held on 26 November at a restaurant in Oslo have been subsequently diagnosed with SARS-CoV-2. More...