Category: Lithuania
Preprint: Long-term mortality following SARS-CoV-2 infection
People infected with SARS-COV-2 had more than three times the risk of dying over the following year compared with those who remained uninfected. More...
West European summer Covid wave brewing *6 updates, 1 prediction*
A new western European summer Covid wave appears to be developing, with increases in cases in countries that also saw large waves in the summer of 2021. More...
Spillover of SARS-CoV-2 across Europe from the war in Ukraine
This is a new post that will be tracking the possible spillover of SARS-CoV-2 from the war zone in Ukraine and into neighbouring countries. More...
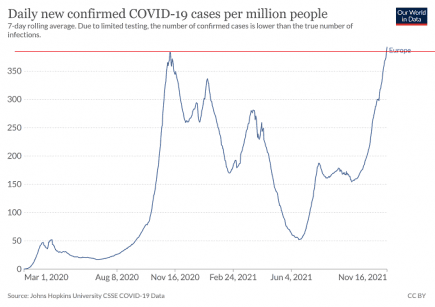
Europe: all-time Covid infection record broken by growing winter superwave
Europe has broken through its all-time high for Covid cases, and is seeing sharp increases in infections right across the continent. More...
Prediction: European Covid winter wave of 2021 will be the worst wave of the pandemic so far
This isn’t the most difficult prediction we have ever had to make, but there is no doubt that, despite huge vaccination campaigns across the continent, the winter wave now breaking over Europe will be their worst wave of the pandemic so far. More...
WHO: 500,000 deaths from Covid in Europe this winter
“The current pace of transmission across the 53 countries of the European Region is of grave concern,” said regional WHO head Hans Kluge. More...
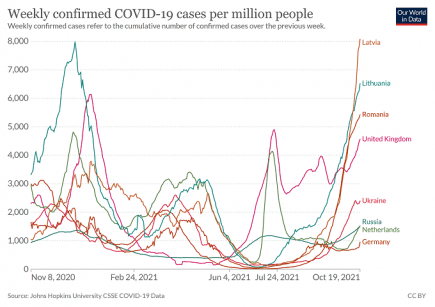
Winter is here. Three European nations see their highest ever Covid case rates
Three European nations, Russia, Latvia and Romania, have recorded their highest ever Covid case rates since the pandemic began, and at least eight European nations are currently seeing their highest Covid case rates for six months. More...
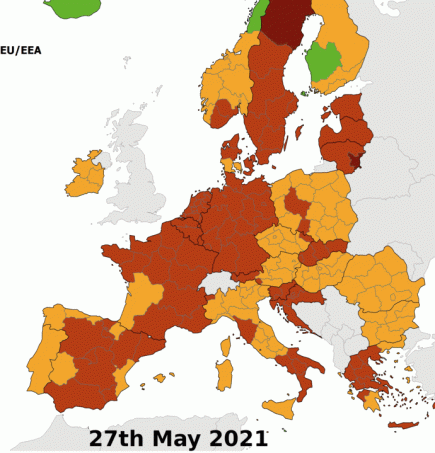
The waxing and waning of the coronavirus pandemic in Europe shows new danger areas emerging
Europe has seen major changes in coronavirus case rates over the last two months. More...
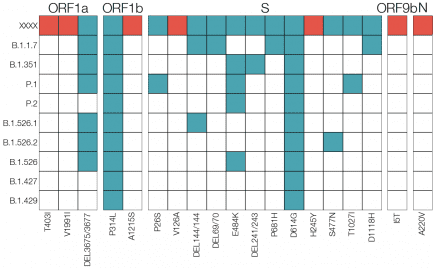
Europe: B.1.620 coronavirus variant – definitely one to watch
“Last month, Gytis Dudas was tracking a concerning new coronavirus variant that had triggered an outbreak of COVID-19 in his native Lithuania and appeared sporadically elsewhere in Europe and in the United States. More...