Category: Side Effects
Geert Vanden Bossche: Mass Vaccination in a Pandemic – Benefits versus Risks
A hugely prophetic video interview with Belgian vaccine developer Geert Vanden Bossche from May 2021, looking at the risks and benefits of vaccinating a population during a pandemic. More...
UK High Court proceedings brought against AstraZeneca for Covid-19 vaccine injuries and deaths
Will the UK High Court finally offer justice to the innocent victims of this god-awful Covid-19 vaccine? More...
Germany: One man, 217 Covid vaccinations
Lancet study: ‘Here, we report on a 62-year-old male hypervaccinated individual from Magdeburg, Germany, who deliberately and for private reasons received 217 vaccinations against SARS-CoV-2 within a period of 29 months.’ More...
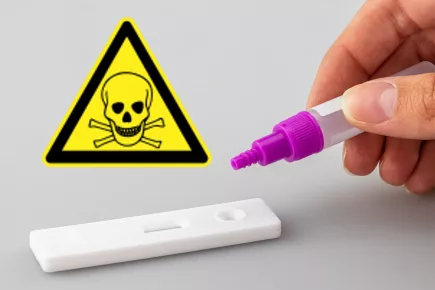
Covid-19 Rapid Antigen Tests Contain Highly Toxic Poison Sodium Azide
‘The vial in many rapid antigen kits includes the chemical Sodium Azide as a preservative agent. More...
U.S. Sen. Ron Johnson roundtable discussion: Contamination of mRNA vaccines with DNA
The latest Covid roundtable discussion, chaired by Senator Ron Johnson, featuring 21 guest speakers. More...
UK NHS healthcare workers launch group litigation against government for Covid-19 injuries received at work
UK NHS workers are preparing to take legal action against the British government for failing to protect them during the Covid-19 pandemic. More...
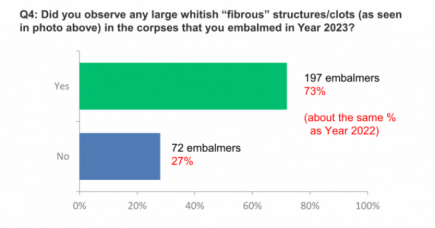
White fibrous clots found in at least 50% of all cadavers embalmed
Another interview by Dr John Campbell, and another Covid-19 bombshell. More...
Tucker Carlson interviews Dr Joseph Ladapo about DNA contamination of Covid-19 mRNA vaccines
The Tucker Carlson Network has released footage of an interview between Tucker Carlson and Dr Jospeh Ladapo, Surgeon General of Florida, exploring the subject of DNA contamination in mRNA vaccines. More...
British Prime Minister finally confronted about Covid vaccine adverse events
Rishi Sunak, the Prime Minister of Great Britain, has found himself on the end of withering criticism over his government’s response to adverse events resulting from the Covid-19 vaccine rollout. More...
Texas Attorney General sues Pfizer over Covid-19 vaccine claims and censorship
The Attorney General of Texas, Ken Paxton, is suing Pfizer for “misrepresenting COVID-19 vaccine efficacy and conspiring to censor public discourse.” More...