UKHSA risk assessment for Omicron variant
UK risk assessment for Omicron variant B.1.1.529, dated 3rd December 2021
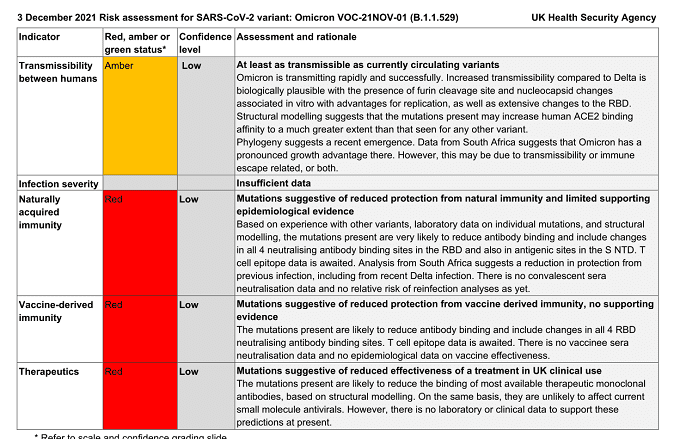
Transmissibility between humans: Amber – At least as transmissible as currently circulating variants
Omicron is transmitting rapidly and successfully. Increased transmissibility compared to Delta is
biologically plausible with the presence of furin cleavage site and nucleocapsid changes
associated in vitro with advantages for replication, as well as extensive changes to the RBD.
Structural modelling suggests that the mutations present may increase human ACE2 binding
affinity to a much greater extent than that seen for any other variant.
Phylogeny suggests a recent emergence. Data from South Africa suggests that Omicron has a
pronounced growth advantage there. However, this may be due to transmissibility or immune
escape related, or both.
Naturally acquired immunity: Red – Mutations suggestive of reduced protection from natural immunity and limited supporting epidemiological evidence
Based on experience with other variants, laboratory data on individual mutations, and structural
modelling, the mutations present are very likely to reduce antibody binding and include changes
in all 4 neutralising antibody binding sites in the RBD and also in antigenic sites in the S NTD. T
cell epitope data is awaited. Analysis from South Africa suggests a reduction in protection from
previous infection, including from recent Delta infection. There is no convalescent sera
neutralisation data and no relative risk of reinfection analyses as yet.
Vaccine-derived immunity: Red – Mutations suggestive of reduced protection from vaccine derived immunity, no supporting evidence
The mutations present are likely to reduce antibody binding and include changes in all 4 RBD
neutralising antibody binding sites. T cell epitope data is awaited. There is no vaccinee sera
neutralisation data and no epidemiological data on vaccine effectiveness.
Therapeutics: Red – Mutations suggestive of reduced effectiveness of a treatment in UK clinical use
The mutations present are likely to reduce the binding of most available therapeutic monoclonal
antibodies, based on structural modelling. On the same basis, they are unlikely to affect current
small molecule antivirals. However, there is no laboratory or clinical data to support these
predictions at present.
