Tag: Astrazeneca
Large UK study confirms previous Covid-19 infection often outperforms vaccine induced immunity
NO, we are ABSOLUTELY NOT recommending you try to get infected with Covid-19! More...
Vaccine waning studies show large declines in efficacy against infection for Covid-19 vaccines
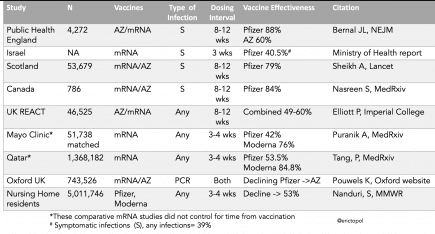
More...A summary table of the studies that have assessed mRNA vaccine effectiveness (VE) for Delta infections, updated with reports from yesterday, overall in line with estimate ~50-60%; *does not pertain* to high VE for hospitalizations or deaths pic.twitter.com/7LxvLoelUI
Pfizer and AstraZeneca vaccine antibodies start to wane after six weeks
Blood tests on hundreds of British people revealed that protective antibodies can wane substantially within weeks of second vaccine shots being given. More...
Wales: Haverfordwest woman’s arm becomes magnetic following coronavirus jab
A HAVERFORDWEST woman says she can now attach small metallic objects to her arm in the area where she had her Covid-19 jab. More...
Bruce Springsteen concert – those with Astrazeneca coronavirus vaccines not invited
Bruce Springsteen has laid down strict rules for attendees of Springsteen on Broadway, which reopens on 26 June at the St James theatre in New York. More...
Human and viral protein impurities found in AstraZeneca’s coronavirus vaccine
“Researchers in Germany report substantial amounts of human and viral proteins in AstraZeneca’s coronavirus vaccine. More...
India: 1.6% coronavirus vaccine breakthrough rate in study of healthcare workers
India’s largest yet data analysis of the so-called breakthrough infections after inoculation with Covishield [AstraZeneca] has found that 16 in every 1,000 fully vaccinated healthcare workers developed Covid-19 infections, about threefold the incidence observed in a US study after Pfizer or Moderna doses. More...
VICTIMS: “Vaccine-Induced Covid-19 Mimicry Syndrome”
FT: “the vaccines send the DNA gene sequences of the spike protein into the cell … Once inside the cell nucleus, certain parts of the spike protein splice, or split apart“
During the last months many countries have started the immunization of millions of people by using vector-based vaccines.
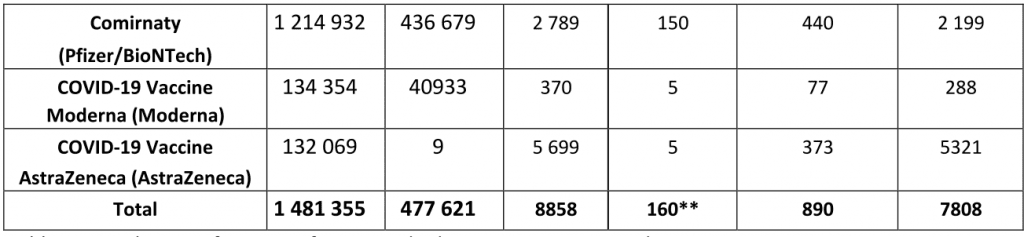
Norway: 1.2 million coronavirus vaccinations, 160 deaths, 890 serious side-effects, 7,800 other side effects
Norway vaccine side-effects to 11th May 2021 (Note: The Astrazeneca Vaxevria vaccine is no longer used in Norway)
Norwegian Medicines Agency report
UK: Eric Clapton had a ‘disastrous reaction’ to AstraZeneca coronavirus vaccination
Photo by Mufid Majnun on Unsplash More...
India: Covaxin and Covishield vaccines invoke 50% fewer antibodies against B.1.617 coronavirus variant
Scientists at the Indian National Institute of Virology (NIV), Pune, have found that both Covaxin and Covishield produce half as many antibodies against the B.1.617 variant of the novel coronavirus as against the ‘original’ B.1 variant. More...