Category: South Korea
North Korea: Omicron BA.2 entered the country on propaganda balloons
“The National Emergency Response Command, as the entry of the malicious virus has been confirmed, is alerting us to weather phenomena, including wind, and unusual objects flying from balloons in the demarcation line and border areas, thoroughly explaining the source, and reporting immediately upon discovery.” More...
South Korea: XE and XM recombinants may be ‘domestic occurrence’
South Korea’s health authority has detected the XE and XM SARS_CoV-2 recombinant mutations for the first time.
Hong Kong: Omicron BA.2 with S:I1221T mutation designated BA.2.2
The dominant BA.2 lineage in Hong Kong with the spike mutation at I1221T has been designated BA.2.2. More...
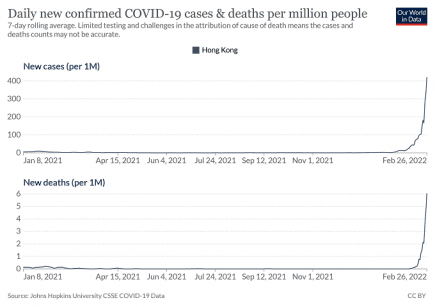
Hong Kong: Link between Covid cases and deaths re-established *4 UPDATES*
Hong Kong may impose a hard lockdown that confines people to their homes, authorities signalled Monday 28th February 2022, with the city’s zero-COVID strategy in tatters and bodies piling up in hospitals. More...
South Korea: 50% vaccine breakthrough for new Covid-19 cases
About half of new COVID-19 cases in the South Korea over the past two weeks were breakthrough infections.
South Korea: Nearly half of recent Covid cases in Seoul are vaccine breakthrough infections
“Of the 646 new cases confirmed in Seoul on Sunday, 49.4 percent, or 319 cases, were breakthrough cases,” Park Yoo-mi, a disease control official at the Seoul metropolitan government, said in a briefing. More...
South Korea: 779 vaccine breakthroughs by vaccine
Janssen’s Covid-19 vaccine recipients reported the largest number of Covid-19 breakthrough infections among the fully vaccinated in South Korea, the Korea Disease Control and Prevention Agency (KDCA) said in a regular briefing on Tuesday. More...
South Korean government temporarily suspends AstraZeneca vaccines for people under 60 after a third blood clot in patient after an AstraZeneca shot
The [South Korean] government is temporarily suspending AstraZeneca vaccines for people under 60 after a third blood clot developed in a patient after an AstraZeneca shot. More...
South Korea: a woman in her 80s died after being vaccinated with AstraZeneca coronavirus vaccine at the Busan nursing hospital
A woman in her 80s who received AstraZeneca Covid-19 vaccine, died in a nursing hospital in Busan. More...
Joint Statement on the WHO-Convened COVID-19 Origins Study signed by 14 countries
The text of the following statement was released by the Governments of the United States of America, Australia, Canada, Czechia, Denmark, Estonia, Israel, Japan, Latvia, Lithuania, Norway, the Republic of Korea, Slovenia, and the United Kingdom.